Biology is a beautiful subject, and once you start to interconnect every topic together, life itself will start to resemble itself as you discover why certain behaviours of organisms are such. I received many requests to make a summary of Units 3/4 biology, so I decided to type of almost everything I know about each topic. I inserted diagrams from various sources to aid the learning process. The information listed below about each topic is exactly how I understand it, and this came from many, many sources from the internet, videos and many different textbooks. Some topics dive too deep into the information, but this was exactly my knowledge in Year 12, so it proves that it does not hurt to know a little bit extra. Please feel free to comment below and leave any suggestions/corrections. Enjoy.
Experimental design
Certain procedures are followed during the course of scientific experiments to ensure that the results are valid.
Hypothesis: - A hypothesis can be created to explain what will occur during the observation
- The hypothesis can be an educated guess or a statement that is predicting a theory that will account for an observation.
- It can be tested and either supported or disproven, but a hypothesis can never be proven.
- The hypothesis should be written as: It is hypothesized that, if
Independent Variable, then
Dependent Variable.
Example:
Independent variable =
red Dependent variable =
green- It is hypothesized that if plants are
exposed to sunlight, then the
rate of photosynthesis will increase.
Variables:- A variable can be any factor, trait or condition that can exist in differing amounts of types. Three different kinds of variables that are referred to: dependent, independent and controlled variables.
- The independent (experimental) variable: This variable is the one that is changed throughout the experiment and does not depend on any other factors. It is best to keep an experiment limited to one independent variable so that any experimental observations can be validly attributed to this.
- The dependent variable: This factor is the one that is changing because of the effect of the independent variable. The change depends on the independent variable. Dependent variable is always measured.
- Controlled variables: Controlled variables are factors that are kept constant for all trials of the experiment.
Controlled experiments:A controlled experiment is composed of two set ups which are the same in every way except for the experimental variable (independent variable). Every other variable, known as the controlled variables, such as sunlight exposure, water availability, food supply, must remain the same between the two set ups. The only thing that should differ is the independent variable. In one set up, the independent variable should be present, whereas in the other set up, it should not be present. After some time, if there are any differences between the two set-ups, then the differences can be validly attributed to the independent variable.
Area of Study 1: Molecules of Life
Biomacromolecules
Macromolecules:There are four main classes of lifes organic compounds that are found in almost all living organisms carbohydrates, lipids, proteins and nucleic acids
The repeating units that serve as the building blocks of a polymer are small molecules called monomers. (Lipids are not polymers).
Synthesis of polymersCondensation reactions (anabolic):
- Monomers are building blocks of larger molecules of the same monomers
- Monomers are joined together by chemical bonds through condensation reactions
- Upon condensation reactions, a molecule of water is released as a by-product
- Condensation reactions are anabolic as two simpler monomers come together to form larger, much complex polymers.
- If two monomers join, this is a condensation reaction.
- If more many monomers join to form a polymer, this is known as condensation polymerisation reaction.
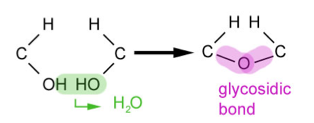 Breakdown of polymers
Breakdown of polymersHydrolysis reactions (catabolic):
- Bonds between monomers can be broken in hydrolysis reactions
- Hydrolysis: the breakdown of a compound due to reaction with water
- This involves the addition of a water molecule to break a longer chain of monomers into its constituent monomers.
 1. Carbohydrates:
1. Carbohydrates:Carbohydrates are composed of saccharide (sugar) monomers and contain the elements carbon, hydrogen and oxygen. The general formula for a carbohydrate is CH2O. This formula applies for simple sugars, such as glucose, and also complex carbohydrates such as glycogen and cellulose.
Carbohydrates are used as a source of energy for organisms. The carbohydrate glucose is vital for cellular respiration.
Carbohydrates can also exist as structural molecules, such as chitin that forms the exoskeletons of insects and cellulose that forms the cell walls of plant cells. Other functions of carbohydrates include existing as a component of the energy transport molecule ATP, as recognition on cell surfaces, and as component of DNA and RNA nucleotides.
- All carbohydrates are composed of carbon, hydrogen and oxygen in the ratio 1C: 2H: 1O
- Larger carbohydrates are broken down into glucose by our digestive system, because a complex polysaccharide is broken down into simpler sugars, this reaction is known as catabolic. The glucose is then absorbed into the bloodstream via villi of the small intestine. Our blood then carries it where it will be absorbed by body cells to be stored as glycogen in the liver and muscle cells, or it can be used for cellular respiration.
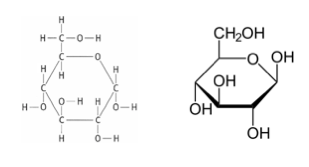
Monosaccharides:- Monosaccharides are the monomers, or subunits, of carbohydrates
- All monosaccharides are soluble in water, that is, they can dissolve in water
- Glucose is the most common sugar that is used in cellular respiration
- Glucose is composed of 6 carbon atoms, 12 hydrogen atoms and 6 oxygen atoms
- C6H12O6 is the general formula for glucose
- Glucose is the monomer that makes up the complex carbohydrates, glycogen, starch, cellulose and chitin
- The structure of glucose:
 Disaccharides:
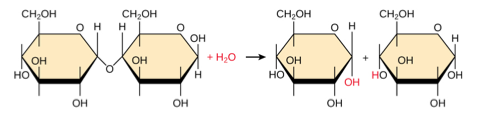
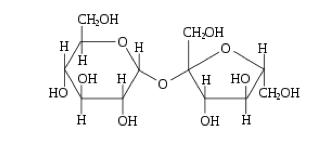
Disaccharides:- Disaccharides are sugars composed of two monosaccharides
- The bond that links two monosaccharides together is called a glycosidic bond. This bond is a strong covalent bond.
- When two monosaccharides link together via condensation reaction, a water molecule is released as the glycosidic bond forms.
- A disaccharide can be split into the two monosaccharides by the addition of a water molecule (hydrolysis). In this situation, water is added and the glycosidic bond breaks to form the original monosaccharides.
 Polysaccharides:
Polysaccharides:- Polysaccharides are not considered to be sugars. Each consists of many sugar units. A polysaccharide is a polymer made of many monosaccharides linked together. The monosaccharides are linked together by glycosidic bonds.
- A series of condensation reactions, known as condensation polymerization, means that many monomers can be joined together to form a polymer.
- There exists four main types of polysaccharides concerned with VCE biology:
Starch: Energy storage in plants
Glycogen: Energy storage in animals in the liver and muscle cells
Cellulose: Structural polysaccharide that is used in plant cell walls to protect plant cells, or supports the shape.
Chitin: Cell walls of fungi and exoskeleton of certain insects.
2.
LipidsLipids are also bio macromolecules, much like carbohydrates, are composed of the elements of carbon, hydrogen and oxygen. The main difference between lipids and carbohydrates in terms of their molecular formula is that lipids contain a greater portion of carbon and hydrogen atoms relative to oxygen atoms.
- Lipids are not polymers because individual lipids are unable to link together to form polymers, as condensation polymerization does not take place.
- Lipids are insoluble in water (they cannot be dissolved in water and hence are hydrophobic)
- Lipids act as energy stores and can insulate the body against heat loss or heat gain.
- Phospholipids also form the backbone of all membranes
- When lipids are broken down during digestion, they are broken down into glycerol and fatty acid sub units.
- Excess lipid, or fat, is stored as adipose in tissue.
- Main functions of certain lipids:
 Fatty acids
Fatty acidsFatty acids are the simplest form of lipid. Each fatty acid is a molecule containing a long chain of carbon and hydrogen atoms (hydrocarbon chain) with a carboxyl group (-COOH) at one end, which is the hydrophilic, water loving or polar section of the lipid.
- There are two types of fatty acids: unsaturated fatty acids and saturated fatty acids
- The main difference between the two is that fatty acids that contain single bonds throughout the hydrocarbon chain have more hydrogen atoms, hence are saturated, whereas fatty acid chains that have a double bond have less hydrogen atoms, and hence are unsaturated.
- Fatty acids are rarely free and by themselves. They are usually found with other types of molecules.
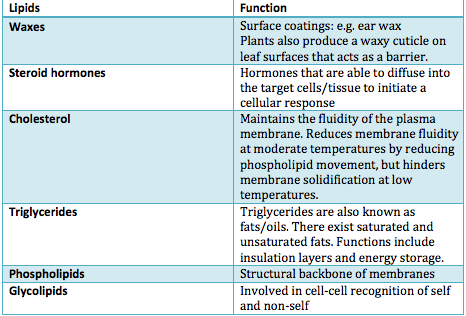
Saturated fatty acids:
- If the fatty acids of a lipid are saturated with hydrogen atoms, then we can conclude that the lipid is saturated with hydrogen atoms; in other words, there are no double bonds between the carbons in the fatty acid chain.
- Saturated fats do not have kinks.
- All saturated fats are solid at room temperature as there are no kinks; saturated lipids are able to closely pack together and hence are found as solids at room temperature.
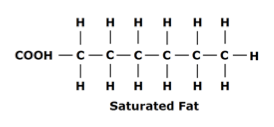
Unsaturated fatty acids:
- If the fatty acids of a lipid are unsaturated with hydrogen atoms, then we can conclude that there exists at least one double bond between the carbons in the hydrocarbon chain (fatty acid chain).
- Unsaturated fatty acid chains have kinks as the double bond causes a slight kink in the structure
- Most unsaturated fats are liquid at room temperature because the kinks (bends) prevent the fat molecules from packing together close enough to solidify, and hence are present as liquids at room temperature.
 Triglycerides:
Triglycerides:Triglycerides lipids composed of a glycerol backbone molecule and three fatty acid chains attached. The glycerol molecule is an alcohol containing three carbon atoms.
Because triglycerides contain fatty acids attached to the glycerol backbone, their state at room temperature is dependent on the nature of the fatty acid chains. If the triglyceride is composed of saturated fatty acid chains, then it will be solid at room temperature. However, if the triglyceride is composed of unsaturated fatty acid chains, then it will be present as liquid in room temperature.
- Triglycerides are a good source of energy in both animals and plants as they contain an abundance of carbon and hydrogen atoms.
- Triglycerides are a good source of insulation.
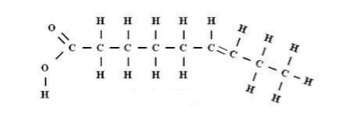
Phospholipids:Phospholipids are a branch of lipids that contain 1 glycerol molecule, two fatty acid chains and 1 phosphate group. Phospholipids are similar to triglycerides, but instead of the three fatty acid chains bonded to the glycerol molecule, a phosphate group replaces one fatty acid chain.
Phospholipids are amphipathic, meaning that they are composed of one part that is hydrophobic, and another molecule that is hydrophilic.
The hydrophobic parts of phospholipids are the fatty acid chains, and are assembled in a bilayer structure and repel away from the aqueous (water) solution that they are suspended in. The hydrophilic parts of phospholipids are the phosphate heads, which are attracted to the aqueous (water) solution and hence face outside the bilayer.
As the phosphate groups are polar and hydrophilic, they are attracted to water in the intracellular fluid. The phospholipid bilayer consists of two adjacent sheets of phospholipids, arranged tail to tail. The fatty acid chains repel from the water, as they are non-polar and hydrophobic, hence associate with each other forming a bilayer as the phosphate heads are exposed to the water. The hydrophobic nature of the fatty acid chains ensures that only lipid-soluble, or non-polar substances can directly diffuse through the cell membrane.
 Glycolipids:
Glycolipids:A glycolipid is a lipid molecule that has a carbohydrate molecule attached to it:
- Glyco = relating to or producing sugar (carbohydrate)
- Lipid = any of a class of organic compounds that are fatty acids or their derivatives and are insoluble in water due to their hydrophobicity.
- The carbohydrates are found on the outer surface of all eukaryotic cell membranes.
- Glycolipids extend from the phospholipid bilayer into the aqueous environment outside the cell where it acts as a recognition site for specific chemicals.
Glycolipids roles are to serve as markers for cellular recognition. They are used to distinguish between self and non-self cells and other foreign material. The different glycolipids on the surface of foreign cells not normally present in the body allow white blood cells to recognize them as non-self, generating the destruction of the invader.
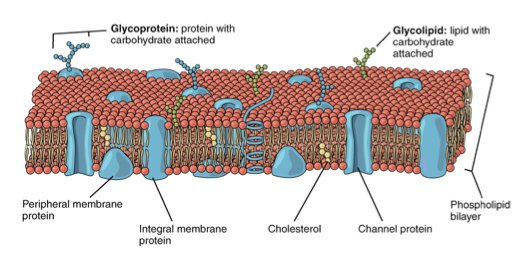
Cholesterol:Cholesterol is a lipid that is found within the fatty acid chains inside the phospholipid bilayer. Due to its lipophilic (hydrophobic) nature, cholesterol is freely found within the bilayer. Its function is to maintain fluidity of the cell membrane of our cells. Under low temperatures, cholesterol prevents the membrane from solidifying, whereas during high temperatures, prevents it from becoming overly fluid. (See image below).
Cholesterol is a lipid and NOT a carbohydrate.
3.
Proteins:The proteome is the complete set of proteins produced by a single cell or an organism. Cellular proteins do not act in isolation from other proteins; in other words, many proteins work in association with other proteins to achieve specific functions in an organism or cell. Scientists study the interrelationships between proteins in a systematic way. This study of the proteome (the complete set of proteins produced by an organism or cell) is known as proteomics.
Proteins are organic compounds, bio macromolecules, which are composed of the following chemical elements: Carbon, Hydrogen, Oxygen and Nitrogen, and occasionally Sulfur. The monomer of a protein is known as an amino acid.
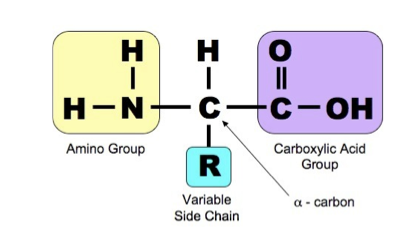
- An amino acid is a relatively small molecule, which contains two reactive groups: an amino group and a carboxylic acid group.
- There are 20 different amino acids that exist in the proteins of humans.
- The difference between amino acids is the different groups of atoms found in the R group.
- R represents the side chain that varies between each type of amino acid.
 Forming polypeptides:
Forming polypeptides:- Polymers of amino acids are called proteins, or polypeptides. These proteins contain many amino acids each bonded through peptide bonds (remember, upon condensation reactions between two amino acids, a water molecule is lost, so the carboxyl group releases the OH and the amino group of an adjacent amino acid loses its Hydrogen atom to form a water molecule, and the bond them forms between C-N).
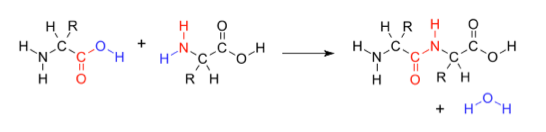
- When two amino acids are positioned so that the amino group of one monomer is adjacent to the carboxyl group of the other, under the presence of an enzyme, these two amino acids can join together to form a dipeptide.
- Di = two
- Peptide = amino acids
- The bond between the C-N is called a peptide bond and a water molecule is released, as this is a condensation reaction.
A protein consists of one or more polypeptide chains folded and coiled into specific shapes. It is the specific amino acid sequence of a polypeptide that determines the final shape of the protein.
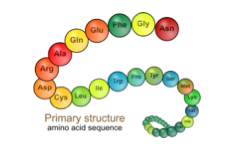
Protein structure:Primary structure- The primary structure of a protein is the specific amino acid sequence.
- There are peptide bonds between these adjacent amino acids
- Even a minor alteration to the primary structure of a protein can have devastating effects on its function and overall structure, that is, if a specific amino acid was replaced by another, this will alter the final protein product.
 Secondary structure:
Secondary structure:- Once the amino acids have been aligned in their specific position, the protein can fold or turn upon it.
- These turns and folds are a result of hydrogen bonding between the functional groups of different amino acids within the polypeptide chain (i.e. the hydrogen bonding occurs between different amine groups and carboxyl groups).
- Two results of the hydrogen bonding occur: Beta pleated sheets and alpha helices.
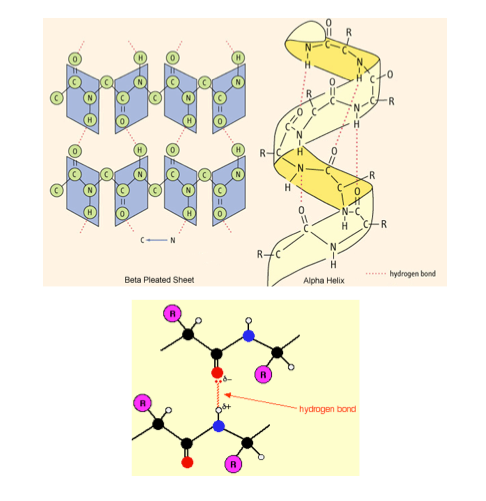 Tertiary structure:
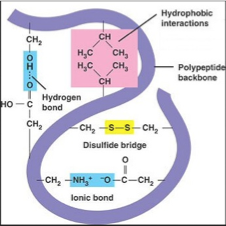
Tertiary structure:- A proteins tertiary structure refers to the way a polypeptide coils to form a complex molecular shape. It is the way in which alpha helices, beta pleated sheets and random coils fold with respect to each other. Irregular bends from bonding between the side chains (R groups) of amino acids in the polypeptide chain create the tertiary structure of proteins.
- The tertiary structure is the overall 3-Dimensional shape as a result of various types of bonding between the different R groups of amino acids within the polypeptide chain.
- Since the function of a protein is dependent on its structure, the tertiary structure is the proteins final structure and hence determines its function.
Some of the R group bonds that may occur include:
- Ionic bonds
- Disulphide bridges
- Hydrogen bonding
- Hydrophobic interactions
- Hydrophilic interactions
Amino acids with hydrophobic (non-polar) side chains are usually found in clusters in the middle of the protein, away from the water. However, hydrophilic side chains of other amino acids are also attracted to each other, but more commonly on the outside of the protein molecule, exposed to the water.
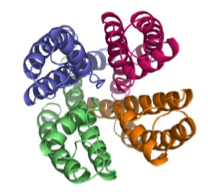
 Quaternary structure
Quaternary structure- When a protein is composed of two or more polypeptide chains, it is known to have a quaternary structure.
 Nucleic Acids:
Nucleic Acids:Nucleic acids are organic compounds that are composed of the following chemicals: Carbon, Hydrogen, Oxygen, Phosphorus and Nitrogen. They store information that determines how organisms develop and function. Nucleic acids are the polymers that are made up of monomers known as nucleotides.
Nucleic acids carry instructions that make proteins by determining the amino acid sequence of the protein produced at the ribosome.
- A nucleotide is the monomer of nucleic acids
- Nucleic acids are either DNA (Deoxyribonucleic acid) or RNA (ribonucleic acid)
- A nucleotide consists of a phosphate group (negatively charged), a sugar group and a specific nitrogenous base.
- Nucleotides can link together through phosphodiester bonds via condensation reactions.
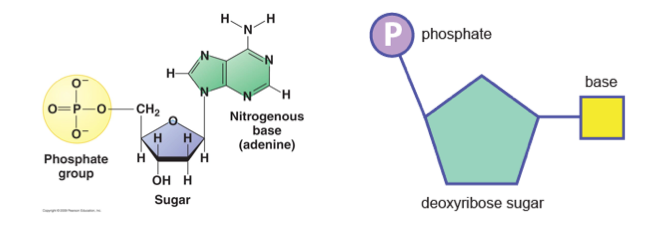 DNA (deoxyribonucleic acids)
DNA (deoxyribonucleic acids)- DNA contains the genetic instructions for all living organisms
- DNA determines the characteristics of every single living organism
- DNA is a nucleic acid, hence its monomer is nucleotides
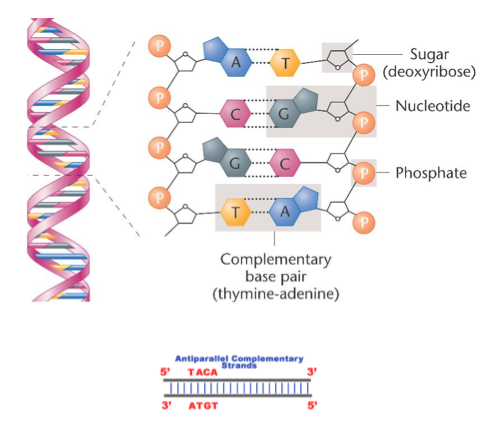
- DNA is a double-stranded molecule (formally known as double helix)
- The sugar molecule of DNA is deoxyribose
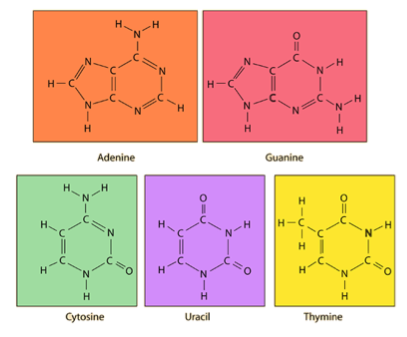
- The Nitrogenous bases in DNA are: Cytosine, Thymine, Adenine and Guanine (GTAC)
- When two DNA molecules align antiparallel to each other, bonds form between the complementary bases of the two strands, forming the double stranded DNA molecule.
- The bonds between nitrogenous bases are hydrogen bonds
- The complementary base pairing rule is as follows: G-C and A-T
- There exists three hydrogen bonds between G and C where as only two hydrogen bonds between A and T
- Adenine and Guanine are known as purines due to their double-ringed structure
- Cytosine and Thymine are known as pyrimidines due to their single-ringed structure
- DNA runs in a direction of 5-3 or 3-5. If the phosphate group is attached to the 5th Carbon on the sugar group, then it is 5 end. If the 3rd Carbon is not bonded to anything, it is 3 end.
 RNA (ribonucleic acid)
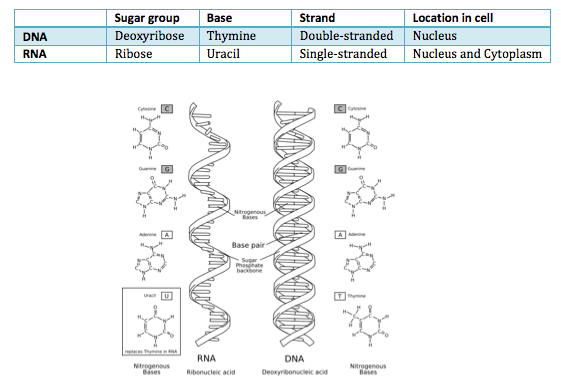
RNA (ribonucleic acid)- RNA is a single stranded nucleic acid, meaning it consists of nucleotides
- The nitrogenous bases of RNA are as follows: Guanine, Adenine, Cytosine and Uracil
- The sugar in RNA is known as ribose.
- There are three types of RNA: mRNA, tRNA, rRNA
The function of mRNA: (messenger RNA)
The mRNA is a large family of RNA molecules that convey genetic information from DNA to the ribosome, where they specify the amino acid sequence of the protein products of gene expression.
The function of tRNA: (transfer RNA)
The tRNA is a type of RNA molecule that helps decode a mRNA sequence into a protein. tRNAs function at specific sites in the ribosomes during translation, which is a process that synthesizes a protein from an mRNA molecule.
The function of rRNA: (ribosomal RNA)
Ribosomal RNA associates with a set of proteins to form ribosomes. These complex structures, which physically move along an mRNA molecule, catalyse the assembly of amino acids into protein chains. They also bind tRNAs and various accessory molecules necessary for protein synthesis.
DIFFERENCE BETWEEN DNA AND RNA:
Plasma membrane
The cell membrane is a very thin structure surrounding every living cell. It is primarily a lipid fluid material, allowing sections of membranes to merge with each other.
The plasma membrane, also known as the cell membrane, is a biological membrane that separates the intracellular fluids from the extracellular fluids of every living cell. The cell membrane also controls the movements of certain substances inside and outside the cell. It consists of the phospholipid bilayer with embedded proteins and other molecules such as: glycolipids, glycoproteins and cholesterol.
As a consequence, a membrane appears like a matrix of substances, hence the structure of the plasma membrane is known as the fluid-mosaic model.
The plasma membrane is semi-permeable, meaning that only selective molecules such as water, ions and other organic molecules are able to pass through the membrane, whereas other molecules are not able to pass through.
The plasma membrane is composed of a phospholipid bilayer. Phospholipids are amphipathic, meaning they have a polar (hydrophilic) and a non-polar (hydrophobic) part. Because of its amphipathic nature, a bilayer exists. The hydrophilic phosphate heads are attracted to water and hence face the outside of the membrane, whereas the hydrophobic fatty acid chains repel and associate with each other inside the bilayer.
Movement of substances in and out of cells:Diffusion: The passive net (overall) movement of molecules from a region of high concentration to a region of low concentration along the concentration gradient until equilibrium is established.
- Diffusion may occur in liquids, such as sugar dissolving in coffee or in water.
- Diffusion across a membrane requires a molecule being transported, or being diffused, across the membrane from a high concentration to a low one.
- This process is passive and does not require energy.
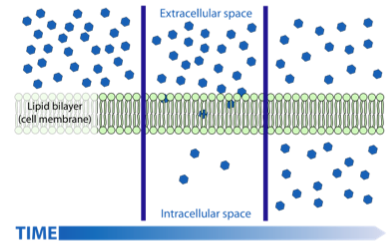
Due to the plasma membranes amphipathic nature (the fatty acid chains = hydrophobic. phospholipids = hydrophilic), large hydrophilic or polar substances are unable to freely diffuse through the membrane, as they repel from the fatty acids. This means that small, non-polar substances such as ethanol are able to freely diffuse across a plasma membrane. However, small particles such as water, oxygen and carbon dioxide, despite being polar and hydrophilic, are small enough to freely diffuse across membranes. Large polar molecules are unable to do so, and hence protein channels and carrier proteins are required to facilitate the movement across the membrane.
Osmosis:- Osmosis is always the diffusion of water molecules across a semi-permeable membrane.
- Osmosis is passive and does not require energy.
- Passive net (overall) movement of water molecules across a semi-permeable membrane from a region of high solvent concentration to a region of low solvent concentration until equilibrium is established.
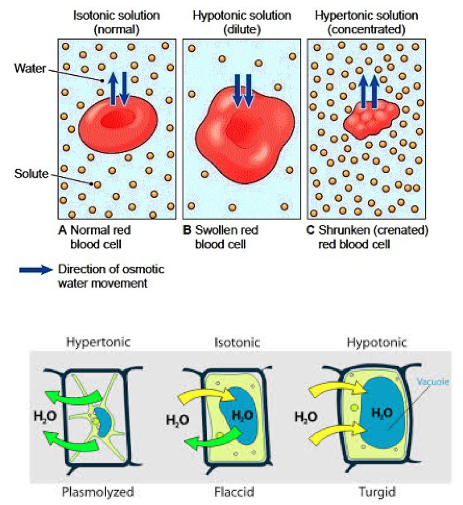
- Hypertonic solution: A solution that has a higher solute concentration than the solution that it is compared with
- Hypotonic solution: A solution that has a lower solute concentration than the solution it is compared with.
- Isotonic solution: A solution that has the same solute concentration as compared with the other solution.
A cell placed in a
hypotonic solution:
- Cells placed in a hypotonic solution means that there is a higher solute concentration inside the cell as compared to the outside solution
- Osmotic gradients exist during osmosis
- Cells placed in a hypotonic solution will tend to gain water as there is a higher water concentration outside the cell, and to naturally reach equilibrium, the water molecules form the out solution diffuse into the cell.
- Animal cells can lyse (burst) when placed in a hypotonic solution if excess water from the solution diffuses into the cell.
- The cell wall of plant cells prevents it fro lysing as it provides strength and supports the shape of plant cells. The cell is termed turgid and exerts turgor pressure instead of lysing.
A cell placed in a
hypertonic solution
- Cells placed in a hypertonic solution tend to lose water through osmosis
- Cells in a hypertonic solution means that the solution they are dispersed in contains a higher solute concentration
- If the solution contains a higher solute concentration than the cell, the water from the cell will diffuse out to establish equilibrium (lower the concentration of the outer solution).
- Hence, cells placed in hypertonic solutions will tend to lose water and crenate (shrivel).
- Animal cells: The process of becoming crenated is known as crenation
- Plant cells: Plant cells become plasmolyzed as the cell membrane retracts from the cell wall, also known as flaccid.
A cell placed in an
isotonic solution
- This means that the solution in which the cells are dispersed into contain the same solute concentration as the cells.
- There is no net movement of molecules as there exists no concentration gradient
- A cell placed in an isotonic solution neither gains or loses water
- Plant cells will remain flaccid (term used to describe their state).
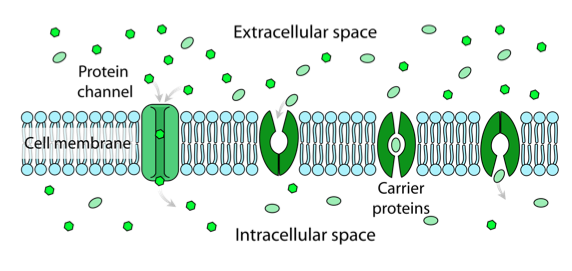
 Facilitated diffusion:
Facilitated diffusion:Facilitated diffusion: is the passive net (overall) movement of a substance from an area of high concentration to an area of low concentration along the gradient concentration via a carrier protein or protein channel in a membrane.
For example: Glucose molecules may pass across membranes in this manner, as it is a large polar molecule.
Two types of proteins are involved in the facilitated diffusion of large water-soluble particles and ions.
-
Channel proteins: these protein channels have a hydrophilic lining and allow relatively larger polar (hydrophilic) substances to diffuse through the membrane.
-
Carrier proteins: these proteins bind to the solute and undergo a conformational change to transport the solute across the membrane.
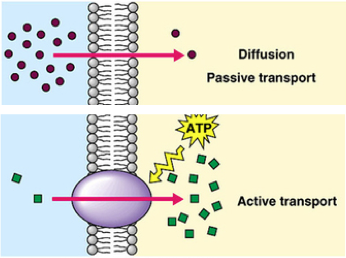
 Active transport:
Active transport:Many substances needed by organisms are required in much greater amounts than can be provided by diffusion alone. Also some substances need to be accumulated in cells against the existing concentration gradient. In both of these circumstances, energy must be used to move the required substances across cell membranes. Such movement is called active transport.
Active transport: the movement of molecules from a region of low concentration to a region of high concentration, using energy in the form of ATP.
Active transport is aided by proteins known as carrier proteins, which have specific receptors that bind to specific molecules and transport them across the membrane, from to the region of higher concentration, and energy is used.
E.g. liver cells up taking more and more glucose for storage as glycogen.
E.g. a sodium-potassium pump in a neuron membrane during resting potential.
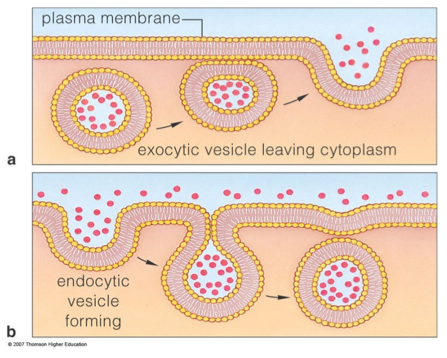
 Active transport: Bulk transport
Active transport: Bulk transportSome substances, including large insoluble particles, can be transported across membranes via two other processes, endocytosis and exocytosis.
Endocytosis: involves the cell membrane sinking inwards, engulfing particles or fluid from outside the cell. A vesicle containing the material pinches off the cell membrane and enters the cytoplasm. This requires energy in the form of ATP.
-
Pinocytosis: liquid brought into the cell
-
Phagocytosis: solid material brought into the cell
The vesicles that are formed by pinocytosis are known as pinosomes, and the vesicles formed via phagocytosis are known as phagosomes. Or in general, they are known as endosomes.
Exocytosis: involves a vesicle, containing particles or dissolved substances, merging with the cell membrane and expelling contents outside the cell. This requires energy in the form of ATP.
Enzymes
Enzymes are protein molecules that catalyse (speed up) chemical reactions. They control rates of reactions in all living organisms. Enzymes are very efficient catalysts for biochemical reactions. They speed up reactions by providing an alternative reaction pathway of lower activation energy.
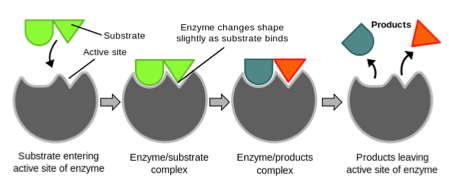
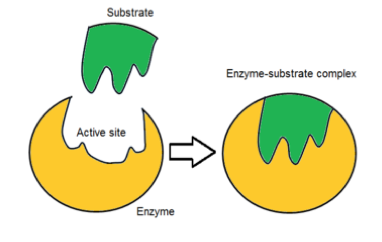
- Active sites on enzymes act on substrates (reactants) and convert them into new products.
- When a substrate molecule binds to the enzyme, an enzyme-substrate complex forms.
- Enzymes are highly specific, as each enzyme typically acts on one substrate only. For example, the enzyme maltase only acts on maltose. This is due to the specific shape of the active site on the enzyme matching the shape of the substrate molecule.
- Most enzymes are intracellular, however, some act outside the cell such as digestive enzymes.
- Enzymes are sensitive to certain conditions in which they can function.
- Specific molecules may alter Enzymatic activity.
- Most enzymes are named after the substrate they act on with the addition of the suffix ase.
 Endergonic and exergonic reactions:
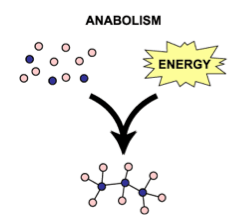
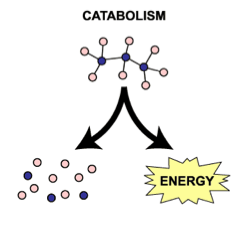
Endergonic and exergonic reactions:Different enzymes are involved in either endergonic (reactions which require energy overall) or exergonic (reactions that release energy) reactions. Enzymes are not destroyed in either reaction, and can be used over and over again.
Endergonic reactions: reactions that require energy and the products have a greater energy level than the reactants.
- This type of reaction is also known as anabolic reactions.
- Anabolic/endergonic reactions involve the synthesis of larger and more complex molecules from simpler molecules.
Exergonic reactions: reactions that release energy.
- This type of reaction is also known as catabolic.
- Catabolic/exergonic reactions involve the breakdown of complex organic molecules into smaller molecules and hence release energy.
 Enzyme action Induced fit or lock and key
Enzyme action Induced fit or lock and keyThere are two theories that describe the action of enzymes, lock and key model and the induced fit model.
Lock and Key: This theory states that the active site of the enzymes fits, exactly, the shape of the substrate it will act on.
Induced fit: This theory states that the substrate induces a change in the shape of the enzymes active site as it binds to the enzyme.
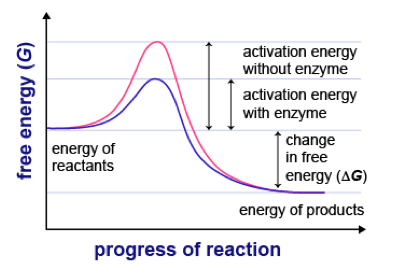
 Activation energy:
Activation energy:Enzymes reduce the activation energy required for a reaction to proceed, and this is why reactions proceed more rapidly.
The temporary association between enzyme and substrate(s) brings reacting molecules closer to one another (during endergonic/anabolic reactions) and weakens existing chemical bonds (during exergonic and endergonic reactions) making it easier for new bonds to form.
 Factors affecting enzyme function:1. Temperature:
Factors affecting enzyme function:1. Temperature: - Each enzyme has an optimum temperature. In humans, this is typically 37 degrees. At high temperatures, enzymes denature, that is, their active sites are destroyed due to changes in the enzymes shape. Hence, enzyme-substrate complexes are no longer able to form, and the enzyme is deemed useless.
- At low temperatures, enzymes become less active due to reduced molecular movement. However, they can regain their former activity again when the temperature rises back to optimum level.
2. pH- Each enzyme has an optimum pH, e.g. pepsin, acting in the stomach, has an optimum pH of 1.5, whereas chymotrypsin in the small intestine works best at a pH of 8.
-
- A change in pH can change the shape of the enzyme active site. Some enzymes have a broad optimal range.
3. Enzyme concentration - Only a small number of enzymes are usually involved in a reaction. If the concentration of the enzyme is increased the rate of reaction will increase.
- The greater the enzyme concentration, the greater the number of enzyme-substrate complexes.
- At high enzyme concentrations, adding more enzyme has no effect on the initial rate of reaction. The rate of reaction is no longer limited by shortage of enzyme but by other factors.
- At low enzyme concentrations, adding more enzyme increases the initial rate of reactions. With more enzymes present, it is more likely that a substance will bind to an empty active site on an enzyme.
4. Substrate concentration - As substrate concentration increases, rate of reaction will also increase (assuming the presence of free enzymes).
Enzyme inhibitors:Certain chemicals (e.g. poisons) can block or alter the shape of enzyme active sites and stop their action.
- Competition inhibitors: have a very similar shape to the substrate molecule, and block the active site to prevent the substrate from forming an enzyme-substrate complex. (They compete for the active site)
- Non-competition inhibitors: attach to another part of the enzyme, resulting in a change in the shape of the active site. (Bind to the allosteric site).
Some common enzymes:
- Maltase: found in saliva and pancreatic juice, acts on maltose.
- Sucrase: acts on the carbohydrate sucrose.
- Caltase: found in all living cells, acts on the metabolic poison hydrogen peroxide, breaking it down to the harmless of by-products water and oxygen.
- Pepsin: attacks food proteins in the stomach
Cellular respiration
Cellular respiration (an exergonic/catabolic reaction)
Cellular respiration involves the breakdown of organic molecules (for example, glucose) and the subsequent release of energy. While most of the released energy is lost as heat, some is trapped in the form of adenosine triphosphate (ATP), and this trapped energy can subsequently be used to power cellular activities.
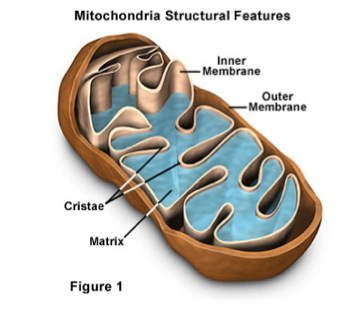
Mitochondria:Mitochondria are the sites of cellular respiration. In the presence of oxygen, they convert energy stored in organic molecules, such as simple sugars and fats, into a form of energy that the cell can use for its many function.
- Many eukaryotic cells contain hundreds of mitochondria. The more active a cell, the more mitochondria it will contain.
- Mitochondria are approximately 1-10um long. Each is composed of two membranes: A smooth outer membrane surrounds a highly folded inner membrane. The infoldings are known as cristae. Cristae contain proteins involved in the electron transport chain or respiration, hence, their large surface area allows for many reactions to take place.
- The fluid surrounded by the inner membrane is the mitochondrial matrix. The matrix is the site of the Krebs cycle, and is rich in respiratory enzymes. The fluid between the inner and outer membrane is the intermembrane space.
- Mitochondria possess their own DNA (mtDNA) and ribosomes.
 Energy and ATP:
Energy and ATP:Sunlight is the ultimate source of energy for all cells. Light energy from the sun is transformed into chemical energy (e.g. in the form of glucose) during photosynthesis. Glucose is an intermediate source of energy for the cell.
An immediate and rapid source of energy, however, is required for cellular activities, and glucose cannot be split quickly enough to release energy for the cells direct needs. Hence, during cellular respiration, when glucose is broken down, and most the energy released is lost as heat, some of the energy is trapped by lurking adenosine diphosphate (ADP) and free phosphate (Pi) molecules to form adenosine triphosphate (ATP), and immediate and transportable source of energy for all cells.
ATP is formed at the site of exergonic (energy releasing) reactions in the cell. its high energy phosphate bond can be split at appropriate times to provide energy for all cell activities.
ATP is formed when ADP reacts with inorganic phosphate. The energy released from carbohydrate (or lipid or protein) during respiration drives this reaction.
When ATPase splits ATP into ADP and Pi, free energy is released for cell activities.
Every living cell is continually respiring, and the rate of respiration varies according to the activity of the cell, e.g. active muscle cells, with numerous mitochondria, can have a very high rate or respiration, while the cartilage cells in your nose, with far fewer mitochondria respire at a much slower rate.
- Cellular respiration involves the breakdown of glucose to release energy
- The complete breakdown occurs via enzyme systems, which operate in the presence of oxygen. The products created are of much lower energy value than glucose, which has had many of its chemical bonds broken.
- Incomplete breakdown of glucose (during anaerobic respiration) results in organic products (e.g. lactic acid) with many of the original chemical bonds remaining intact, i.e. less energy has been released.
Aerobic respiration
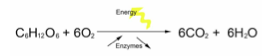
- Aerobic respiration: Involves the breakdown of glucose into simple inorganic compounds (carbon dioxide and water) in the presence of oxygen
Aerobic respiration involves many steps, many enzymes and many vitamins that act as coenzymes. Respiration involves a series of enzyme-controlled reactions:
Overall molecular equation:
Aerobic respiration can be divided into three main stages:
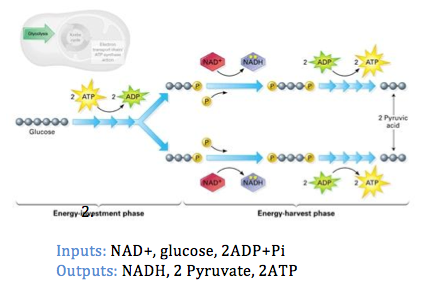
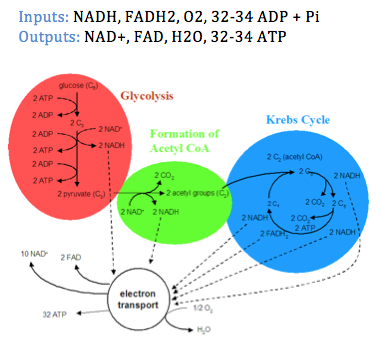
1. Glycolysis: - Occurs in the cytosol of cells
- A glucose molecule is partially broken down into two 3-carbon molecules (pyruvates).
- Hydrogen ions are removed from glucose in this process. These hydrogen ions are used in the formation of loaded acceptor molecules (NAD+ + H+ -> NADH)
- Energy released from the breakdown of glucose into two pyruvate results in the formation of 2ATP molecules.
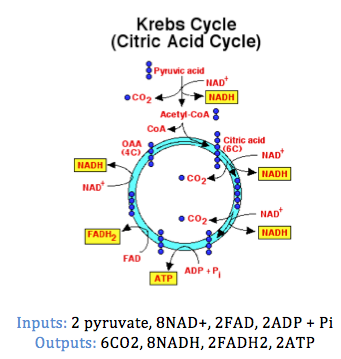
 2. Krebs cycle:
2. Krebs cycle: Occurs in the mitochondrial matrix (fluid surrounded by inner membrane) or mitochondria. The two pyruvate molecules produced in glycolysis enter the mitochondria. The decomposition of each pyruvate will result in the formation of three molecules of CO2.
- Within the mitochondrion, pyruvate is converted to acetyl CoA (One CO2 molecule released and one NADH released).
- Acetyl CoA enters the Krebs cycle, and during this cycle another 2 CO2 molecules are released. Note: Overall 6CO2 molecules released, as 3 per pyruvate and 6 per glucose molecule.
- The hydrogen ions released during the Krebs cycle are used in the formation of loaded acceptor molecules NADH and FADH2.
- Altogether, 2ATP molecules are released from the breakdown of the two-pyruvate molecules.
 3. Electron transport chain:
3. Electron transport chain: Most of the ATP produced during respiration occurs when the loaded acceptor molecules produced by the Krebs cycle (NADH and FADH2) pass the electrons originally extracted from food molecules (e.g. glucose) to the electron transport chain.
- Occurs in the cristae of mitochondria (the inner projections of the inner membrane).
- Electrons removed from food are transferred by NADH to first electron carrier molecule of electron transport chain
- Note: Electron carrier molecules are embedded in the cristae membranes.
- As electrons pass from one carrier molecule to the next, energy is released in manageable amounts, resulting the formation of 32-34 ATP molecules.
- This cytochrome passes the electrons to oxygen, which also picks up a pair of hydrogen ions, thus forming water.
Anaerobic respiration
- In conditions where oxygen is absent or very limited, cells can still respire through anaerobic respiration pathways. Many organisms use such pathways all the time.
- Organisms that typically rely on aerobic respiration, however, can also supplement their energy requirements by utilizing anaerobic pathways, particularly if the oxygen supply for aerobic respiration is insufficient to meet the energy demands of the cell.
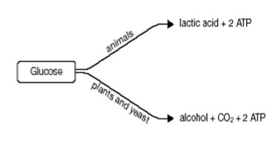
- Anaerobic respiration: Involves the breakdown of glucose to smaller organic compounds in the absence of oxygen. As there is incomplete breakdown of glucose, less energy is released per glucose molecule as compared with aerobic respiration.
- First stage (glycolysis): in the cytoplasm, glucose is split into two pyruvate molecules, and 2ATP molecules are also formed.
- Second stage in animals (Lactic acid fermentation): In the cytoplasm, pyruvate is converted to lactic acid and water, with no release of CO2.
- Second stage in plants, yeast (alcohol fermentation): Pyruvate is converted into ethanol and carbon dioxide in the cytoplasm.
- In animals, once the oxygen supply is again sufficient to meet the energy demands of the cell, the excess lactic acid is converted back to pyruvate, which may then enter the Krebs cycle or be anabolised to glucose for storage.
- In plant and yeast cells, the accumulated ethanol cannot be reconverted back into pyruvate or glucose. Ethanol is toxic in cells; hence, anaerobic respiration can only be performed for short periods of time in plant and yeast cells.
Photosynthesis
Photosynthesis involves the synthesis of organic molecules (e.g. glucose) from water and carbon dioxide using energy absorbed by chlorophyll from sunlight. Light energy is converted to chemical energy during this process, and as with respiration, it involves a series of enzyme-controlled reactions.
Overall worded equation: Carbon Dioxide + Water -> Glucose + Water + Oxygen
Overall chemical equation: 6CO2 + 6H2O -> C6H12O6 + 6O2
Photosynthesis occurs in plant and protists cells containing chloroplasts (as well as in cyanobacteria with free-floating chlorophyll). Chlorophyll absorbs light energy and converts it to chemical energy (in the form of sugars) in a series of many enzyme-controlled reactions. It mainly absorbs the red and blue-violet wavelengths of the light spectrum, and reflects most green lights.
Chlorophyll, the principle pigment required for photosynthesis, is found in the thylakoid membranes of chloroplasts. Chlorophyll maximally absorbs the blue/violet and red wavelengths of the light spectrum.
Factors affecting photosynthesis:- Carbon dioxide concentration (because carbon dioxide is an input, if there is limitations to this reactant then it will affect the rate)
- Light intensity
- Water availability
- Temperature
- Oxygen concentration
- Chlorophyll concentration
Chloroplasts:Chloroplasts are specialized plastids found in many plant and protists cells. They contain the green pigment chlorophyll, and are the site of photosynthesis in all eukaryotic cells.
- Chloroplasts are found in leaves and other green regions of plants, as well as in algae.
- As with mitochondria, the chloroplast contains two membranes, however, both membranes are smooth without folding.
- A narrow intermembrane space exists between the two membranes. Inside the inner membrane, another membrane system (thylakoid membrane system) exists. In some sections the thylakoids are stacked like green pancakes, forming structures called grana.
- The fluid surrounding the grana is called the stroma.
- As with mitochondria, chloroplasts possess their own DNA and ribosomes and can divide in two, as well as being mobile organelles.
The two stages of photosynthesis
Light dependent and light independent stagesLight dependent:- The reactions occur in the grana (thylakoid membranes)
- Light energy is absorbed from the sun light via chlorophyll
- Water molecules split to form H+ ions and O2 gas. The O2 gas is released via the stomata. Excited electrons flow through the electron transport chain to provide energy for ATP synthesis
- Unloaded electron acceptor molecules, NADP+, accept H+ ions to form NADPH
Inputs: H2O, ADP + Pi, NADP+
Outputs: O2, ATP, NADPH
Light independent:- Occur in the stroma of chloroplasts
- In the Calvin cycle, CO2 and H+ ions (from the NADPH formed in the light dependent reactions) are used to synthesize sugars.
- Energy is provided by the ATP molecules produced during the light dependent reactions
Inputs: CO2, ATP, NADPH
Outputs: Glucose, ADP+Pi, NADP+
Glucose is the main product of photosynthesis, however, other organic molecules are also produced:
- Glucose and fats can be used as energy sources in the process of respiration
- Cellulose is synthesized from glucose monomers to form the cell wall
- Proteins have a myriad of functions within the cell.
Note: Oxygen is produced as a waste product of photosynthesis and is released from the chloroplast during the light dependent reactions.
Summary of PHOTOSYNTHESIS:
Photosynthesis is the process whereby sunlight (radiant) energy is converted into chemical energy via a pathway of enzyme-controlled reactions. Two main stages of photosynthesis are involved and are known as the light dependent reaction and light independent reaction. The light dependent reactions require sun light, the sun light is captured by the chlorophyll pigments on the thylakoid membranes and this energy is sued to split water into hydrogen ions and oxygen gas. Oxygen is released as a by-product and the hydrogen ions occupy unloaded acceptor molecules to form NADPH and ATP. These NADPH and ATP molecules now exit the thylakoid membranes and flow into the stroma, where they undergo the light independent reaction. Carbon dioxide is reacted with these molecules to produce an organic molecule, glucose, and excess hydrogen ions from the NADPH are used to form water molecules.
Area of Study 2:
Detecting and Responding
Homeostasis and Multicellular organisms.
Detecting and responding involves communication between the cells of an organism and the organism and its environment. This involves:
- Production of a signal
- Detection of the signal
- Transfer of the signal
- A response to the signal by the organism
- Switching off the signal
- If the communication is faulty in any of the above pathways, an inappropriate response, disease or even death may occur.
Homeostasis: Involves processes that are responsible for maintaining a relatively constant internal environment in response to change/stimulus.
The feedback responses:Homeostasis involves the operation of feedback mechanisms, whereby the response affects the original stimulus. It is important that the response be controlled or turned off once conditions have returned to normal levels. This reduces energy wastage by the organism.
- Negative feedback mechanisms are very common in the body and involve two stages. A stimulus or a change is detected and then a response is produced to oppose the stimulus or return the original stimulus back to normal levels.
- If the stimulus-response mechanism has been adequate in cancelling the stimulus, the stimulus-response cycle no longer continues.
- The control center for transmission is usually a part of the CNS, such as the hypothalamus.
Hormones and other chemical messengersHormones are organic chemical messengers produced and secreted from specific cells that travel via extracellular fluid and act on the specific target cells, causing an appropriate cellular response. Only target cells possess receptors for the specific hormone.
After their secretion, most mammalian hormones travel in the bloodstream to exert their effects in a different area of the organism. Some chemical messengers however, act on the same cell that produced them, and others are secreted into the surrounding tissue fluid only to act on neighboring cells.
- Chemical messengers, hormones, are found in both plants and animals.
- Despite being relatively slow acting in nature, hormones can exert long lasting effects in the organism.
There exist
three main types of hormones:
- Protein hormones: insulin, glucagon, adrenalin
- Steroids (lipids): testosterone, estrogen, cortisone
- Amino acid derivatives: thyroxin
- In the case of protein/peptide or amino acid derivative hormones, the hormone-secreting cell typically releases its contents via exocytosis. Steroid hormones and some amino acid derivative molecules, however, pass our of their hormone-secreting cells by simply diffusion.
- Hormones attach to receptors on or in target cells via lock and key mechanisms. The hormone attaches to a receptor site (typically a protein molecule) that is only found in target cell tissues. If a cell does not have the required receptor site, then the hormone will not bind with the cell and hence no cellular response will be initiated.
- Receptor proteins are found in two locations. There are transmembrane receptors that are found on the membrane of cells, and there are cytoplasmic receptors which are found inside cells.
- Transmembrane receptors form hormone-receptor complexes with protein based hormones as these hormones are large and polar and hence unable to diffuse through the cell, so they bind with receptors on the cell surface
- Steroid hormones are able to diffuse into the cell, and hence bind with cytoplasmic receptors inside the cell
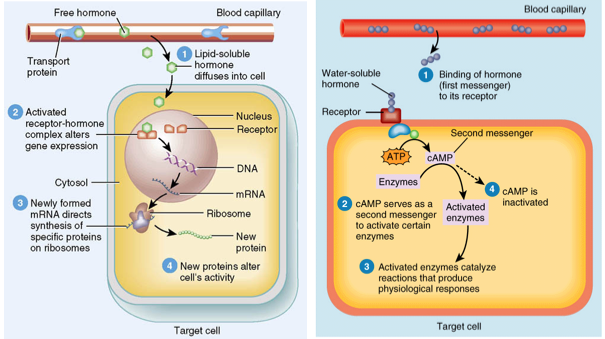
Action of water-soluble and lipid-soluble hormones:Based on their mode of operation, hormones can be divided into two broad groups, water-soluble and lipid-soluble hormones.
Water-soluble hormones: - Protein hormones
- Peptide hormones
- Most amino acid derivative hormones
- In the case of water-soluble hormones, the receptors are located on the surface of the cell membrane of the target cells.
- The hormone-receptor complex activates other proteins within the cell, secondary messengers, (e.g. G proteins) which are responsible for the cascade of events (signal transduction) within the cell leading to the cellular response.
These hormones dissolve readily in water, but are unable to cross membranes.
Lipid-soluble hormones: - Steroid hormones
- Some amino acid derivative hormones (e.g. thyroxin)
- Lipid hormones are not soluble in water, and can freely pass through membranes.
- In the case of lipid-soluble hormones, the receptors are found within the target cell, either within the cytosol or the nucleus. Again, the hormone binds to the waiting receptor. The hormone-receptor complex attaches to a particular site on a chromosome within the nucleus, leading to the cells response.
Whether or not a hormone is water or lipid-soluble,
the target cell must contain protein receptor molecules for that particular hormone. Summary of cell signaling:
Cell signaling occurs in this manner:
1. Signal reception
2. Signal transduction (read below)
3. Cells response
Signal transduction:Signal transduction at the cellular level refers to the cascade of events leading to a specific cellular response.
- With each step in a signal transduction pathway, the number of molecules involved is typically amplified; many more molecules are typically involved at each subsequent step in the pathway. The outcome is an alteration in cellular activity, which can be the result of changes in gene expression within the cell.
First and second messengers:The extracellular signal molecule that binds to the membrane receptor is the first messenger. Many signaling pathways involved small, typically non-protein, and water-soluble molecules of ions second messengers. As second messengers are both small and water soluble in nature, they can rapidly spread throughout the cell by diffusion, thereby amplifying the cells response.
Functioning of a steroid hormone receptor:1. Lipid-soluble steroid hormone diffuses through the membrane.
2. The hormone binds to the cytoplasmic protein receptor
3. The hormone-receptor complex then enters the nucleus
4. The hormone-receptor complex binds to specific regulator sites for the targeted gene (activator or repressors. In this case, activators for step 5 and 6).
5. Stimulating the gene to produce mRNA
6. mRNA is read by ribosomes to produce a specific protein
Summary:
a. Water soluble hormone:
Binds with transmembrane receptor
The hormone-receptor complex initiates a second messenger
The second messenger triggers the signal transduction pathway a cascade of reactions
A specific cellular response is carried out
b. Lipid soluble hormone:
- Diffuses straight through the cell membrane due to its lipophilic nature
- Either binds to cytoplasmic receptor or receptor in the nucleus
- The hormone-receptor complex activates gene expression
Apoptosis
In a healthy organism, cell death is balances by cell production (via mitosis). Programmed cell death can vary greatly. The majority of blood cells age and will die. Each cell generally has a maximum number of cell divisions it can undergo before its programmed death. The cell ceases to perform cellular processes and dies.
Apoptosis is the process of programmed cell death that may occur in multicellular organisms. Biochemical events that lead to characteristic cell changes and death. These changes include cell shrinkage, nuclear fragmentation, chromatin condensation, and chromosomal DNA fragmentation.
Fragmentation: the process or state of breaking or being broken into fragments
- Apoptosis is programmed cell death. It is not the death of a cell due to damage of tissues (necrosis)
- Every second, more than a million of our cells divide by mitosis, so to maintain the correct number of cells in the body, over one million cells must activate enzymes (caspases) which digest the cell from within every second of our life.
- Dying cells display signals recognized by macrophages, which engulf the cell remnants.
- Some cancers are caused by genes, which prevent cell death by stopping caspases from being activated.
The following steps take place during Apoptosis:
- Caspases are activated; dying cells release signals to arouse phagocytes
- Cells begin to shrink and develop small bumbs on their surface
- Caspases enter nuclear pores to chop up DNA and proteins; mitochondria break down
- Small fragments bind to receptors on phagocytes are engulfed
- Phagocytes release cytokines to prevent inflammation and neighboring cell death.
Immune system
The immune system is the collection of organs and processes of the body that provide resistance to infection and toxins. Organs include the thymus, bone marrow, and lymph nodes.
There are
three lines of defence:
1. First line of defence
2. Second line of defence
3. Third line of defence
First line of defence:The first line of defence includes physical and chemical barriers that are always ready to defend the body from infection, non-specifically. These include your skin, tears, mucus, cilia, stomach acid, urine flow, bacteria and white blood cells called neutrophils.
Examples of physical or chemical first line defence barriers:
- Intact skin: provides non-specific protection by disallowing foreign materials to enter body
- Ciliated membranes: hair-like projections trap foreign substances and waft the particles up to the throat, where they are either coughed or sneezed out.
- Natural flora: Naturally occurring non-pathogenic bacteria inhabit our skin and prevent other pathogens from entering through competition
- Stomach acid: kill foreign pathogens
- Tears: dehydrate foreign pathogens, as tears are high in solutes.
- Mucus: lining traps foreign material
- Saliva: Contains antibacterial enzymes
Second line of defence and third:- If pathogens are able to surpass the first line of defence, for example when a cut appears on your skin and an infection appears, the second line becomes active.
- The second line of defence involves structural (cells) and processes that work non-specifically to destroy invading pathogens.
- Cells involved in second line of defence are known as white blood cells, leucocytes. Each of these leucocytes has a specific function, but they all work together to protect us.
Summary of Specific Immune Response:Bacterial cells may infect an organism, humans. These bacterial cells will attract phagocytes and will be engulfed. Once engulfed, antigens are presented on their MHC II markers to activate T cells. Once T cells are activated, they go and activate B cells, and the cytotoxic T cells will regulate the body. The B cells must form an antigen-antibody complex and engulf that complex, present antigens on MHC II markers, and TCRs of the T helper cell must bind with those MHC II markers in order for B cell proliferation. These B cells will proliferate into B plasma cells, which secrete and produce antibodies specific for the antigen, and into memory B cells, which are stored in the spleen and lymph nodes for long term immunity. Immune responses involving antigens and antibodies are commonly referred to as specific, and the non-specific reactions will defend the organism in the same way for every pathogen.
PDF:
https://www.dropbox.com/s/gl9utdl222mxrvg/Cosine%27s%20Biology%20Megathread%20%28Unit%203%29.pdf?dl=0
