Can someone help with this question?? Probably simple but I've forgotten 
Hey! This is a tiny area in the Semiconductor section, and it's just something you need to memorise. When you dope a semi-conductor with a Group 3 metal (ie. P-type), an extra band called the
acceptor band is formed slightly above the valence band. When you dope a semi-conductor with a Group 5 metal (ie. N-type), an extra band called the
donor band is formed slightly below the conduction band. These are totally arbitrary notions, and only make sense when you physically draw the levels. Both of these things makes it 'easier' for the electrons to jump between the valence and conduction bands, making it easier to conduct a charge!
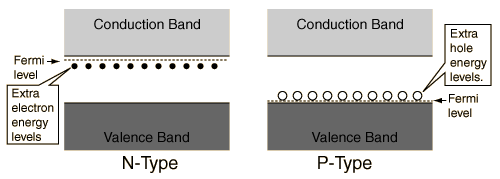 Above courtesy of Hyperphysics, a great online resource!
Above courtesy of Hyperphysics, a great online resource! So, to your actual question. Based on the location of the additional band, we're expecting a P-type semiconductor. This means it has been doped with a Group 5 element. The answer, therefore, is Boron.
Quick note; you could have quickly eliminated half of the potential solutions (B and D) noting that we can't dope semi-conductors with those elements. Then, you had a 50-50 chance of getting the question right, even if you had no idea what it was even asking!Hope this is clear. It's a totally arbitrary section of the curriculum (and of Physics), so don't worry if you forgot how it works!
Jake

